Overview
Syngene, a leading global contract research organisation (CRO)/contract development and manufacturing organisation (CDMO), partnered with a US-based rare disease company to develop and manufacture a first-in-class treatment for a rare disease affecting children.
Syngene successfully developed the manufacturing process for the drug and supplied the required quantities to assist the company during different phases of drug development (preclinical and clinical), and later for commercial manufacturing.
The company received authorisation for the sale of the product in the European Union (EU), followed by regulatory approvals for sale in the US and UK. With this, the product has become the first drug in the world to be approved for children suffering from the rare disease.
The challenge
The company decided to partner with Syngene for process development and manufacturing of their product after developing it for several rare paediatric diseases. We received the discovery process for the synthesis of the product and set about developing the manufacturing process to improve the throughput and quality. During this process, we encountered the following challenges:
- Need to redefine the regulatory starting material (RSM) strategy during the late stage of the development process
- Need for effective planning and execution to bring all cross-functional teams onto a single platform for successful process validation
- Need for an in-depth technical understanding of the process and analytical methods to ensure flawless process validation
- Need for prompt troubleshooting support by all cross-functional teams to meet stringent project timelines
- Working during the Covid-19 pandemic, with all its restrictions such as social distancing and mask wearing, without affecting project timelines
- Need for a smooth transition to Syngene’s new digital systems (to ensure business as usual during a pandemic), including effective, first-time implementation in line with project requirements

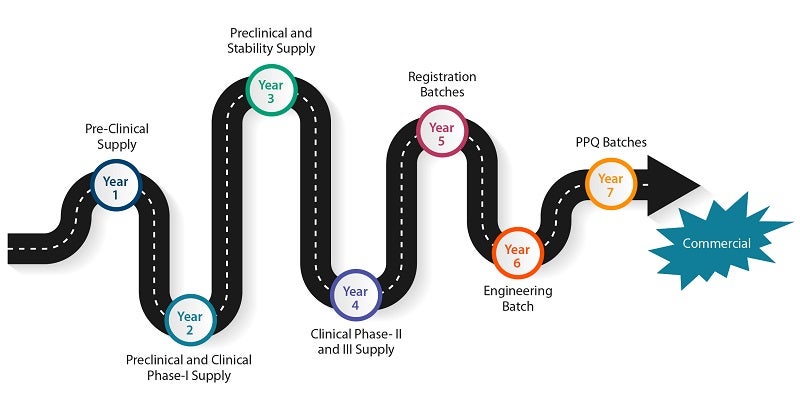
Product journey from lab development to drug substance validation
Syngene played a pivotal role in the development of the product. The company required assistance in product development across different phases (pre-clinical and clinical). We manufactured and supplied the required quantity of the product in conjunction with the process development team.

Syngene’s approach
Defining strategies for designating RSMs
The product’s journey started with the development of appropriate regulatory starting materials (RSMs).
We took up this challenge by undertaking process development studies on Syngene premises. This was followed by analytical method development and stability studies. Later, based on the recommendations of the regulatory authorities, we came up with a strategy for designating the RSMs.
We quickly developed a process for the defined RSM and identified reliable vendors for the procurement of other RSMs. We then implemented this strategy for manufacturing registration batches of the product.
Process validation strategy
Based on our success with registration batches, we redefined the strategy for performing process validation (PV) or process performance qualification (PPQ) batches for the product.
A focused chemical development team was deployed to work on this program to enable the PPQ batches. During this period, the team successfully completed pre-validation studies for the process and analytical methods. Following this, we completed the key phase of the programme – engineering and process validation campaigns for the product.
PV or PPQ validation is required to assure stakeholders that the processes will produce consistent and repeatable results within the predetermined specifications. PV also verifies whether the quality standards and compliance requirements are being met by the product in real time – an important consideration for any pharmaceutical facility.
To accomplish the PPQ campaign, we deployed a dedicated chemical development team from various cross-functional areas. The campaign enabled the gathering of complete and in-depth information about control strategies, critical process parameters (CPPs), critical quality attributes (CQAs), scale-up parameters, and so on. We also undertook detailed pre-validation studies and assessment of existing campaign knowledge to ensure the success of the campaign.
Performing pre-validation studies
A substantial commitment is required to perform pre-validation studies, both in terms of time and resources. Another major requirement is to work in line with ICH guidelines to provide a defined and robust quality control strategy, including identifying CPPs and unit operations that impact any defined CQAs.

| Activities/Studies | Outcomes |
|---|---|
| Existing process and knowledge assessment | Process development summary report |
| Process and analytical method development | Process validation and analytical method validation |
| Identification and characterisation of impurities | Synthesis of impurity reference standards and markers |
| Spike purge/process control justification (PCJ) studies | Control strategies and critical quality attributes (CQAs) |
| Process characterisation, including design of experiment (DoE) | Robustness, critical process parameters (CPPs), normal operating range (NORs), proven acceptable range (PARs) |
| Analytical method (AM) verification, including precision, accuracy, limit of quantification (LOQ), limit of detection (LOD) | Analytical method validation, mechanism of action |
| Process engineering and process safety studies | Equipment qualification, safety risk assessment |



At Syngene, process validation also involves collecting and evaluating data from the process development stage through to commercial production. This is done to establish the scientific evidence that a process is capable of consistently delivering quality products, including assurance of the reliability of supply. Based on successful pre-validation studies, we defined the process validation master plan and initiated the process validation campaign for the product.
Integrated approach to process validation
We used an integrated approach to carry out the process validation campaign, which required expertise across various disciplines. This included the process research and development (PRD) team/analytical research and development (ARD) team, the scale-up team comprising technology transfer, process engineering and production teams, and the quality management team (quality control and quality assurance).
To ensure the success of the programme, we prepared a detailed plan involving effective coordination with cross-functional teams and support from senior management. We successfully completed PV/PPQ batches along with pre-validation data with the defined CPPs and CQAs. This helped the company submit the final new document application (NDA) documents to the US Food and Drug Administration (FDA) and European Medicines Agency (EMA) regulatory authorities.
NDA submission to regulatory authorities
The next step was to obtain the pre-approval inspection (PAI) for the drug substance. Syngene played a significant role in helping the company complete this process successfully.
Following the PAI, the company submitted a marketing authorisation application (MAA) to EMA and an NDA to the US FDA seeking approval for the product.
The EMA granted the product accelerated assessment and orphan designation, as well as access to the PRIority MEdicines (PRIME) scheme. Similarly, the programme received fast-track, rare paediatric disease and orphan drug designations in the US.
The product has since been approved in the US, EU and other markets.
Conclusion
Syngene’s association with the biotech company only goes to show how outsourcing critical services to a reliable partner can help accelerate drug development programmes.
We not only helped to define RSMs for the product but also successfully developed the manufacturing process to improve the throughput and quality of the product.
Further, in conjunction with process development, we manufactured and supplied the required quantity of the product for preclinical and clinical supply and, more recently, for commercial manufacturing. We also provided support for submitting marketing applications for the product in various regions.
As a result of our combined efforts, the product has become the first drug in the world to be approved for children suffering from the rare disease.
To know more about our drug development services or to contact our experts, please click here.


