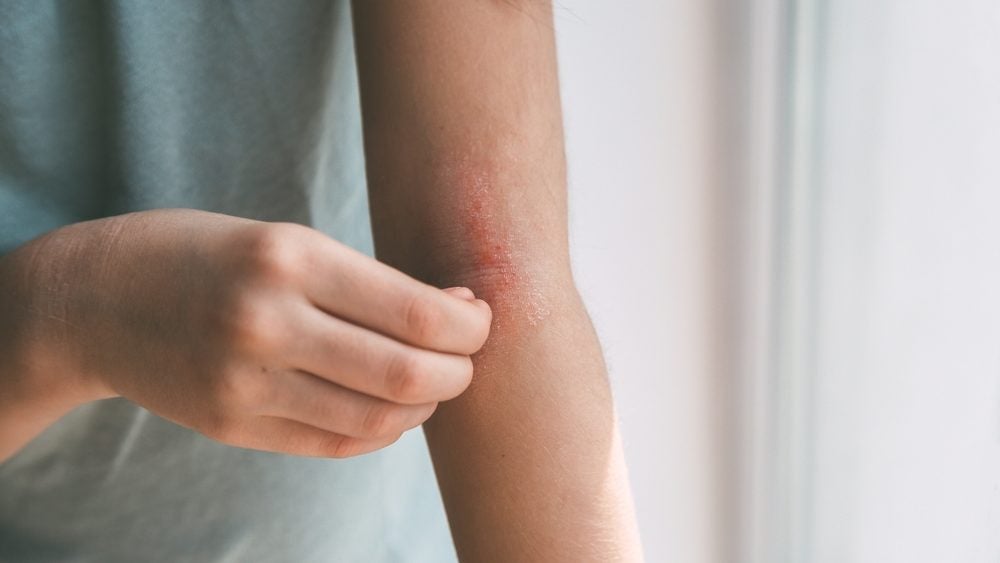
Arcutis has won its third US Food and Drug Administration (FDA) approval for Zoryve (roflumilast) cream in two years, with the latest indication meant for adults and some children with atopic dermatitis.
The FDA has approved the once-daily topical cream for mild to moderate atopic dermatitis in patients aged six years and older. Zoryve, a phosphodiesterase-4 (PDE4) inhibitor, was approved as a cream in July 2022 for the treatment of plaque psoriasis for patients aged 12 and older, and October 2023 for children aged 6-11 with psoriasis.
The initial date for the label expansion in atopic dermatitis was set for 7 June 2024, but the decision was delayed by several days, with Arcutis releasing a statement on 9 June saying there had been no request for new information from the regulator at the time.
The approval is supported by positive data from three Phase III studies, a Phase II dose-ranging study, and two Phase I pharmacokinetic studies. Pooled data from the Phase III INTEGUMENT-1 and INTEGUMENT-2 studies were announced by Arcutis in January 2024. At the four-week mark, 91.5% of patients treated with the drug for atopic dermatitis experienced a measurable improvement in the Eczema Area and Severity Index (EASI) score after four weeks.
Topical steroids have been the gold standard for atopic dermatitis treatment for the last 50 years. However, Zoryve offers a new option for patients without some of the risks associated with topical and systemic steroids. Atopic dermatitis is a chronic skin condition characterised by dry, itchy, and inflamed skin. According to GlobalData, the atopic dermatitis market is forecast to generate $24.4bn by 2030 in the seven major pharmaceutical markets (US, UK, France, Spain, Italy, Germany, and Japan).
GlobalData is the parent company of Pharmaceutical Technology.
This label expansion may drive increased sales for Zoryve. The Zoryve portfolio – which includes the cream and foam formulations– generated $21.5m for Arcutis in Q1 2024, as per the company’s financials. According to GlobalData’s Pharma Intelligence Center, Zoryve is forecast to generate $582m in sales in 2030.
Another non-steroidal cream in the space is Incyte’s JAK inhibitor Opzelura (ruxolitinib), which gained approval for atopic dermatitis in 2021. However, since Opzelura needs to be applied twice daily, and only covers adults and children aged 12 and over, Zoryve may hold an advantage. Opzelura made sales of $86m in Q1 2024 for its labelled use in across atopic dermatitis vitiligo.
Arcutis’ CEO Frank Watanabe said: “Zoryve is the fastest-growing steroid-free topical, relied on to provide effective and safe results in any location on the skin for any duration. With the addition of the new 0.15% strength of Zoryve cream for AD to the higher-strength cream and foam products, the Zoryve portfolio has the potential to become the preferred topical brand in dermatology.”